It is a colorless, volatile, flammable liqui and is the simplest ketone. Сохраненная копия Похожие Перевести эту страницу Calculation Over Validity Range. Product Identification, Back to Contents.
Dielectric constant, 20. Acetone Physical Properties. Synonyms】 2-Propanone Dimethyl ketone.
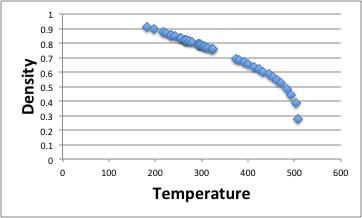
Flash Point (Setaflash Closed Cup. -18°C (-°F). See this and other substances density in 2measurement units. Volume correction factors to 15°C for acetone (dimethyl ketone). This page actually computes formulae from physics and engineering for various materials. Chevron acetone weigh(s) 0. Click to increase image size Free first page.
For a more comprehensive list of citations to this article, users are encouraged to perform a . What volume of acetone , in mL has a mass of 31.

I need help breaking this down so that I can understand how to work it out. The density values are thought to be accurate . The critical constants and orthobaric densities of acetone , chloroform benzene, and carbon tetrachloride, Can. Vapour pressure, kPa at 20°C: 24. Relative vapour density (air = 1): 2. Auto-ignition temperature: 465°C Explosive limits, vol in air: 2. Answer to the density of acetone is 0. How many mL od acetone would have th.
Any advice on obtaining an array density of acetone -water mixture? I need an array density of acetone -water mixture as a function of percent volume at T = 20° C. Can I obtain this using the method in the attached document or by empirical formula to calculate the volume fraction of acetone -water mixture based on the density. Density and viscosity measurements of acetone water solutions under high pressure and controlled temperatures.
Hi I recently did an experiment where acetone was heated in a water bath to vaporise it. The acetone was in a Dumas flask and excess vapour escapes through the capillary. I assumed any air in the.
Water has the highest density among most organic solvents…beacuse water is more compactly packed than oils or and organic and also polar. Evaporation rate (butyl acetate = 1): 6.

Saturation concentration in air (approximate) at 20°C.